|
Thalamic Stimulation
During physiologic localization in preparation for thalamotomy, the observation that high-frequency stimulation of the ventral intermediate nucleus of the thalamus abolished tremor led to investigation of thalamic stimulation as a treatment for tremor. The first study of this technique as a long-term therapy for tremor was reported in 1993.19
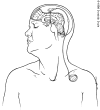
Thalamic stimulation involves implanting an electrode in the thalamic area found to be responsible for the tremor. After the wire of the electrode leaves the skull, it is tunneled under the scalp and down the neck to a purse generator located in the subclavicular pouch (Figure 1). The implanted stimulating device is much like a modified pacemaker, and its electrical impulses can suppress tremor indefinitely. The stimulator can be reprogrammed by using a small portable computer that communicates with the device by radio frequency. Moreover, the patient can turn the device on and off with a magnet. Patients usually turn the device on in the morning, leave it on during waking hours and turn it off at bedtime, since most tremors cease during sleep.
In the first study of this technique,19 as many as 88 percent of the patients with Parkinson's disease had either good or excellent relief of tremor. The operative risk of implanting the device is proving to be similar to that of thalamotomy; death, paralysis, aphasia and significant cognitive deficits are possible complications.
Tremor recurrence after placement of the electrode can be controlled by adjusting the stimulation parameter rather than by reoperation.20 The U.S. Food and Drug Administration has approved thalamic stimulation as an accepted therapy for unilateral suppression of uncontrolled essential tremor or parkinsonian tremor in an upper extremity. As with the other surgical techniques, thalamic stimulation is an option that should be chosen only after medical therapy has failed.
Promising Surgical Approaches
At the forefront of new surgical therapies for tremor are pallidal stimulation and subthalamic nucleus stimulation.21-23 With new advances in deep brain stimulation, procedures can be performed bilaterally to relieve tremor in patients with bilateral involvement. Either a combination of thalamotomy and stimulation or bilateral stimulation without ablation is now a possibility.23 Targets in the brain that are too dangerous to approach for producing a lesion by means of thalamotomy may be treated with stimulation instead, and electrical stimulation can be modified to alleviate tremor as it progresses.22 Thus, deep brain stimulation has become a promising option for abolishing tremors that cannot be controlled by medical therapy.
The authors have received honoraria from Allergen, Inc., Medtronic, Inc., Roche Laboratories, SmithKline and Beecham Pharmaceuticals, and Upjohn Company, and research support for clinical trials from Allergen, Inc., Roche Laboratories and Medtronic, Inc.
The authors thank Dominick Doyle, Medical Arts Group, Vanderbilt University, Nashville, Tenn., for providing the drawing in Figure 1.
The Authors
P. DAVID CHARLES, M.D.,
GREGORY J. ESPER, B.S.,
iTHOMAS L. DAVIS, M.D.,
ROBERT J. MACIUNAS, M.D., DAVID ROBERTSON, M.D.,